Media Matters: Serum-Free Hematopoietic Cell Culture for Gene Editing and Gene Therapy Research
SPOTLIGHT ON CELL THERAPY RESEARCH
Scientists developing new cell therapy methods for the treatment of disorders such as sickle cell disease or leukemia need to consider how their culture conditions may affect the success of their translational research.
Hematopoietic Cell Therapy: Where are we now?
Hematopoietic stem cells (HSCs) have long been used to provide lifesaving treatments for patients with malignant and non-malignant hematological disorders. While originally these treatments took the form of allogeneic bone marrow transplants, more recent breakthroughs in the field have provided cell therapy researchers with methods for correcting genetic mutations in a patient’s own cells to provide an autologous source of HSCs for transplant, or to expand cells ex vivo and extend the potential of allogeneic cell sources. The variety of new tools available for genetic manipulation now include lentiviral-based gene delivery, and gene editing using CRISPR/Cas9, zinc finger nucleases (ZFNs) or transcription activator-like effector nucleases (TALENs).
In order to modify or expand primary human hematopoietic cells, they must first be isolated from the body and then cultured under conditions that allow for their modification. Recent papers published by groups from UC Berkeley and Stanford University have described methods for the genetic manipulation of primary human hematopoietic stem and progenitor cells (HSPCs) that require them to be maintained in vitro for anywhere from 24 hours to more than a week.
Culturing HSPCs in Serum-Free Media for Cell Therapy Research
Whether a protocol uses cell culture methods to expand edited cells for downstream assays, to increase the efficiency of lentiviral transduction or to let cells ‘rest’ after electroporation, optimized culture conditions are key to maintaining cell viability and biological function.
StemSpanâ„¢ serum-free medium undergoes rigorous QC testing to ensure consistent conditions for the culture of HSPCs. StemSpanâ„¢ media do not contain cytokines or growth factors, allowing researchers the flexibility to establish cultures that meet their specific requirements.
As compared to other serum-free media, StemSpanâ„¢ SFEM II medium supports the superior expansion of CD34+ cells when combined with cytokine supplements formulated specifically for this purpose (Figure 1).
StemSpanâ„¢ and Gene Editing
Gene editing has emerged as a promising strategy to treat diseases like β-thalassemia and sickle cell disease which are both caused by mutations in the gene for β-globin (HBB).
CRISPR-Cas9 has been explored by several groups and recent publications by DeWitt et al. [1] and Dever et al. [2] demonstrate a great potential for this technology to one day help patients in the clinic. Cell culture steps were used by both groups to let cells recover after thawing and/or electroporation, and additionally to generate erythroid progenitor cells from edited CD34+ cells[1]. This research shows great promise as a therapy not only for β-hemoglobinopathies, but also for a range of other hematological diseases.
StemSpanâ„¢ and Gene Therapy
Lentiviral-based gene therapy methods to modify human CD34+ hematopoietic stem cells have been investigated as a way of treating various hematological disorders including X-linked chronic granulomatous disease (X-CGD). X-CGD is caused by a genetic abnormality that results in the impaired production of antimicrobial compounds by neutrophils. Consequently, patients with X-CGD suffer from severe infections and excessive inflammation.
Recently De Ravin et al. [3] published their work using ZFNs to provide therapeutic transgenes in an effort to compensate loss-of-function mutations in X-CGD. This group cultured human CD34+ cells in StemSpanâ„¢ medium and cytokines as a pre-stimulation step ahead of electroporation, before xenotransplantation into mice or additional in vitro culture. They applied this method to modify HSCs isolated from patients with X-CGD and found that this restored the antimicrobial activity of neutrophils derived from the transgene-containing cell population.
StemSpanâ„¢ and Ex Vivo Expansion
In addition to bone marrow, cord blood is another potent source of HSCs for transplantation. Cord blood provides several benefits over bone marrow, including the need for less stringent HLA-matching, though its utility can be limited by the availability of fewer HSCs in each sample. In an effort to extend the use of cord blood transplants to more patients, researchers are looking to identify specific molecules and culture conditions which allow for the ex vivo expansion of HSPCs before transplantation.
Delaney et al. [4] who have shown that cord blood-derived CD34+ cells cultured in StemSpanâ„¢ medium with cytokines, on plates coated with Notch ligand, are expanded ex vivo while still maintaining their ability to engraft into immunocompromised mice. Since then, Delaney et al. [4] have progressed to clinical trials to observe the effect of these ex vivo expanded cells in patients.
In 2014, Fares et al. [5] discovered the small molecule UM171 as a novel stimulator of the ex vivo expansion of cord blood-derived CD34+ cells. By culturing cells in a fed-batch system with StemSpanâ„¢ medium containing cytokines and UM171, expanded cells were found to successfully engraft and repopulate immunocompromised mice, with no disadvantage when compared to unmanipulated cells.
Superior Ex Vivo Expansion of CD34+ Cells in StemSpanâ„¢
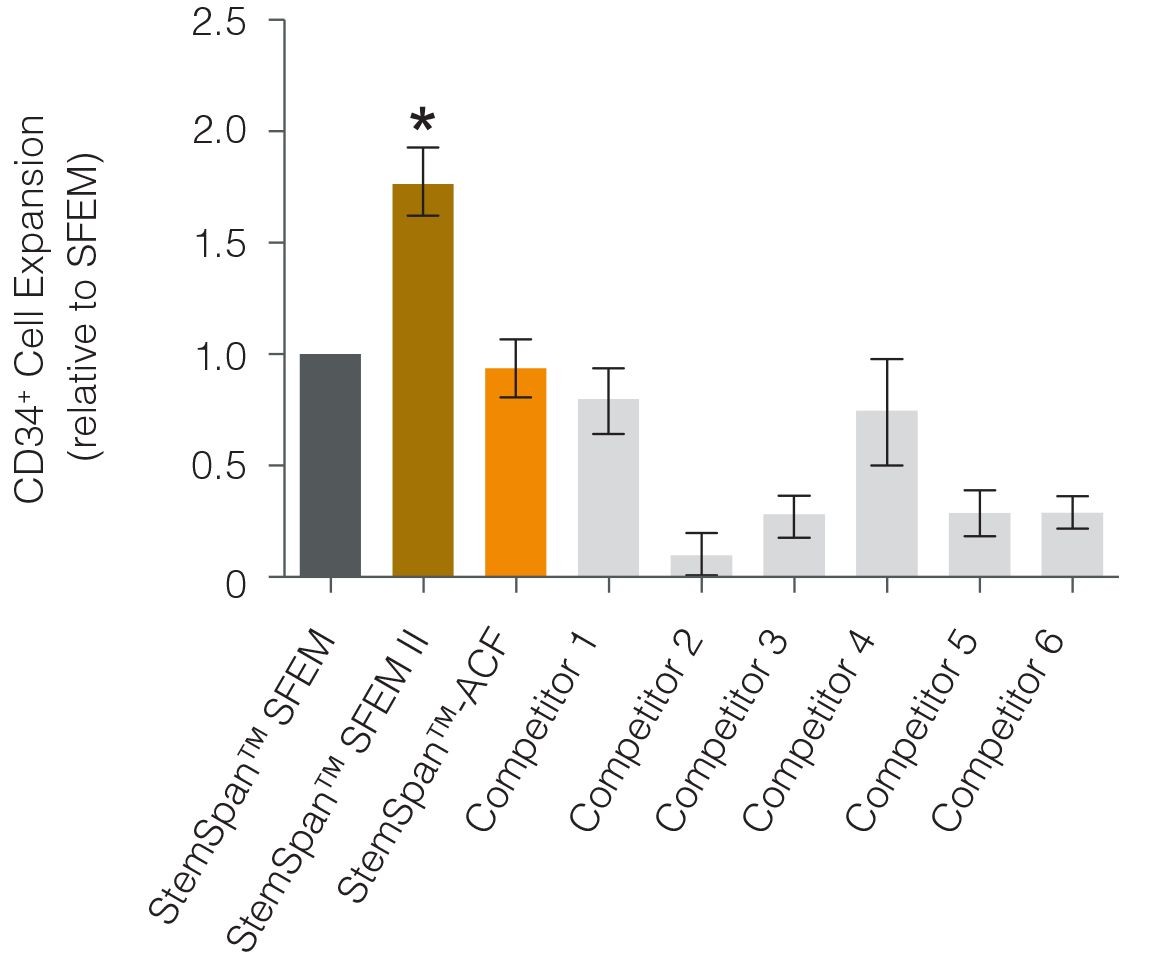
Figure 1. StemSpanâ„¢ SFEM II Serum-Free Expansion Medium Containing CD34+ Expansion Supplement Supports Greater Expansion of Human CD34+ Cells Than Other Media Tested
Expansion of CD34+ cells normalized relative to the values obtained in SFEM medium (dark gray bar) after culturing purified CD34+ cord blood cells for 7 days in StemSpanâ„¢ serum- [SFEM, SFEM II (gold bar)] and animal component-free ACF (orange bar) media, and six media from other suppliers (light gray bars). All media were supplemented with the StemSpanâ„¢ CD34+ Expansion Supplement. Vertical lines indicate 95% confidence limits, the range within which 95% of results fall. The numbers of cells produced in StemSpanâ„¢ SFEM II were significantly higher than in all other media (*p<0.01, paired t-test, n=6).
For more information, check out our recent webinar with Dr. Suzan Imren on her work with Drs. Colleen Delaney and Guy Sauvageau on ex vivo culture and expansion of normal and leukemic hematopoietic cells. You can learn more about StemSpanâ„¢ for serum-free HSPC expansion and more tools for HSPC expansion research on our website. For any other questions please contact us.
References
1. DeWitt MA, Magis W, Bray NL, Wang T, Berman JR, Urbinati F, Heo S-J, Mitros T, Muñoz DP, Boffelli D, Kohn DB, Walters MC, Carroll D, Martin DIK, Corn JE. Selection-free genome editing of the sickle mutation in human adult hematopoietic stem/progenitor cells. Sci Transl Med, 8(360): 360ra134 (2016)
2. De Ravin SS, Reik A, Liu P-Q, Li L, Wu X, Su L, Raley C, Theobald N, Choi U, Song AH, Chan A, Pearl JR, Paschon DE, Lee J, Newcombe H, Koontz S, Sweeney C, Shivak DA, Zarember KA, Peshwa MV, Gregory PD, Urnov FD, and Malech HL. Targeted Gene Addition to a Safe Harbor locus in human CD34+ Hematopoietic Stem Cells for Correction of X-linked Chronic Granulomatous Disease. Nat Biotechnol, 34(4): 424—429 (2016)
3. Delaney C, Heimfeld S, Brashem-Stein C, Voorhies H, Manger RL, Bernstein ID. Notch-mediated expansion of human cord blood progenitor cells capable of rapid myeloid reconstitution. Nat Med, 16(2): 232-6 (2010)
4. Fares I, Chagraoui J, Gareau Y, Gingras S, Ruel R, Mayotte N, Csaszar E, Knapp DJ, Miller P, Ngom M, Imren S, Roy DC, Watts KL, Kiem HP, Herrington R, Iscove NN, Humphries RK, Eaves CJ, Cohen S, Marinier A, Zandstra PW, Sauvageau G. Cord blood expansion. Pyrimidoindole derivatives are agonists of human hematopoietic stem cell self-renewal. Science. 345(6203):1509-12. (2014)
5. Dever DP, Bak RO, Reinisch A, Camarena J, Washington G, Nicolas CE, Pavel-Dinu M, Saxena N, Wilkens AB, Mantri S, Uchida N, Hendel A, Narla A, Majeti R, Weinberg KI & Porteus MH. CRISPR/Cas9 β-globin gene targeting in human haematopoietic stem cells. Nat Biotechnol, 539(7629): 384-389 (2016)