MSCs in research: current trends and future developments
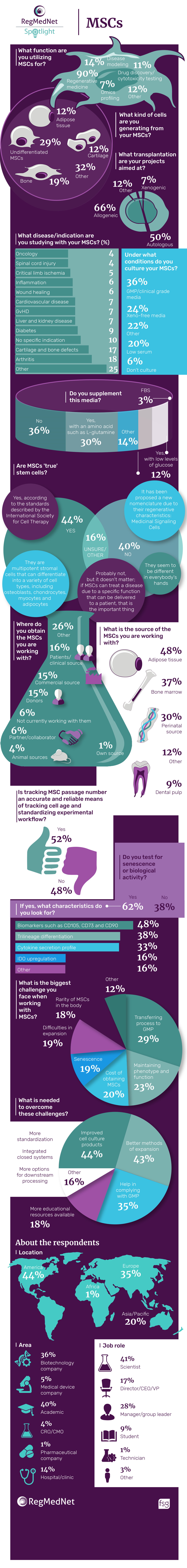
In this infographic, we present the results of our latest spotlight survey on MSCs.
In our latest spotlight survey, we wanted to find out how and why you are utilizing MSCs in your research. How do you compare? Tell us in the comments below or on Twitter at @RegMedNet.
In our latest community post, Melissa van Pel and Brigitte Wieles (Leiden University Medical Center) share their thoughts on the results.