Are your hPSC cultures what you think they are?
In this interview, Melanie Kardel and Adam Hirst (STEMCELL Technologies, BC, Canada) share expert tips on ensuring hPSC quality for your research.
Please introduce yourself and STEMCELL Technologies.


We are both Senior Scientists at STEMCELL Technologies, developing products for human pluripotent stem cell (hPSC) maintenance, characterization and for facilitating innovative techniques, such as gene editing, in hPSCs.
Melanie completed her PhD from the University of British Columbia (BC, Canada) where her research focused on hPSCs and their differentiation into hematopoietic cells. Adam completed his PhD from the University of Sheffield (UK) where he focused on genomic instability in hPSCs.
STEMCELL develops specialized cell culture media, cell isolation systems, accessory reagents and scientific services that are used by scientists working in the cell therapy, cancer research and regenerative medicine fields. STEMCELL also partners with world-leading researchers to commercialize discoveries and help advance life sciences research around the globe.
What unique challenges are involved in performing research on human pluripotent stem cells?
Melanie Kardel (MK): Human pluripotent stem cell research is such a rapidly evolving field that it can be hard to keep up with new discoveries and protocols. For researchers joining the field, wading through the existing protocols to pick the best ones can be overwhelming when nearly every publication has its own unique tweaks. Culture methods themselves have become simpler over the years, but they still require specialized techniques and labor-intensive routine observation and feeding. The lengthy workflows create a lot of potential bottlenecks that can introduce selection or variability.
Adam Hirst (AH): A major concern for the hPSC field is reproducibility of data between labs–a contributing factor to this is the sheer number of cell lines being generated and used in studies globally. Reprogramming has become increasingly accessible and efficient, with labs able to generate a number of cell lines rapidly. With no consensus on validation and characterization of induced pluripotent stem (iPS) cell lines, even different clones from the same reprogramming experiment can have different growth characteristics or differentiation potentials.
What are the most common pitfalls you think new researchers to the field should be aware of?
MK: Since the field is moving so fast, it can be tempting to dive right into a new project after generating iPS cell lines without taking the time to bank and characterize these cells appropriately. Characterization of banks should include the basics, such as assessing morphology and marker expression, as well as identity testing, functional demonstration of pluripotency and genetic testing for karyotypic abnormalities. Prioritizing cell quality early saves time, money and effort, as you will avoid running into surprises at the end of a project. Plus, if issues do arise, you will be able to safely return to that original bank of characterized cells.
AH: Rapid advances in gene editing and organoid technologies have made hPSCs a tool for researchers outside of the PSC field. Particularly with new hPSC researchers, it is easy to overlook the fact that getting to know your cell lines is important, as being able to identify changes in culture quality characteristics such as morphology, growth rates, cell death and spontaneous differentiation levels can provide earlier indications of potential issues.
With many procedures involving multiple steps, which could be stressful for cells, what control measures should researchers take to ensure that their cells are of high quality downstream?
MK: Starting with well-characterized, high-quality input cells is definitely the most important thing–you can’t get high quality cells downstream if you don’t have them upstream. Researchers should be mindful of events that could cause bottlenecks in cell survival, for example during cloning. Minimize stress during these bottlenecks by using optimized culture conditions, such as using our CloneR™2 supplement, and monitor for changes in cell quality characteristics following these stressors. We’ve compiled some of the tools that we use to monitor cell quality on our website.
It is also important to avoid stressful situations on a daily basis, for example, medium acidosis indicated by yellowing culture medium. Even with an enhanced medium like mTeSR™ Plus, this may require feeding dense cell cultures with increased medium volumes or more frequently, or splitting the cultures before they get too confluent.
How often, or at what points in the process, should researchers be assessing cell quality?
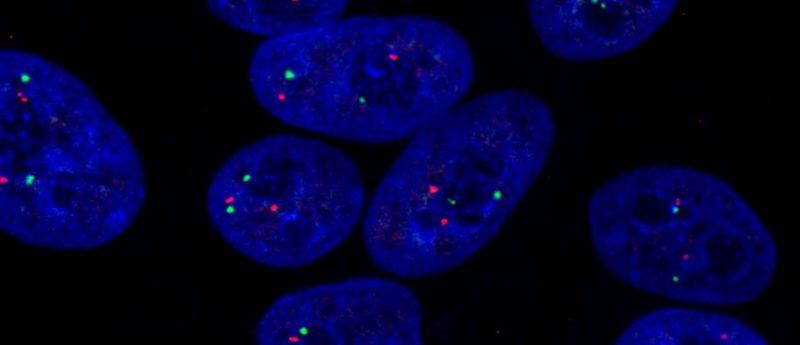
AH: Cell quality should be monitored throughout the culture process. What varies is the level and type of assessment carried out. Documenting culture quality characteristics and media color are quick assessments that can be done daily and highlight issues early. I would also recommend assessing genetic integrity at regular intervals, for example monthly, to monitor cell cultures for potential abnormalities. The hPSC Genetic Analysis Kit was developed for this application; it provides a simple, fast and cost-effective method to screen cell lines.
The more in-depth testing that Melanie mentioned for new cell banks should be performed when a new iPS cell line is generated or line is cloned following gene editing, and at the end of a study to demonstrate that the cells have not changed. It is also good practice to thaw fresh cells from the bank at regular intervals (10-20 weeks) during your experiments to reduce the length of time and number of passages that cells experience in culture.
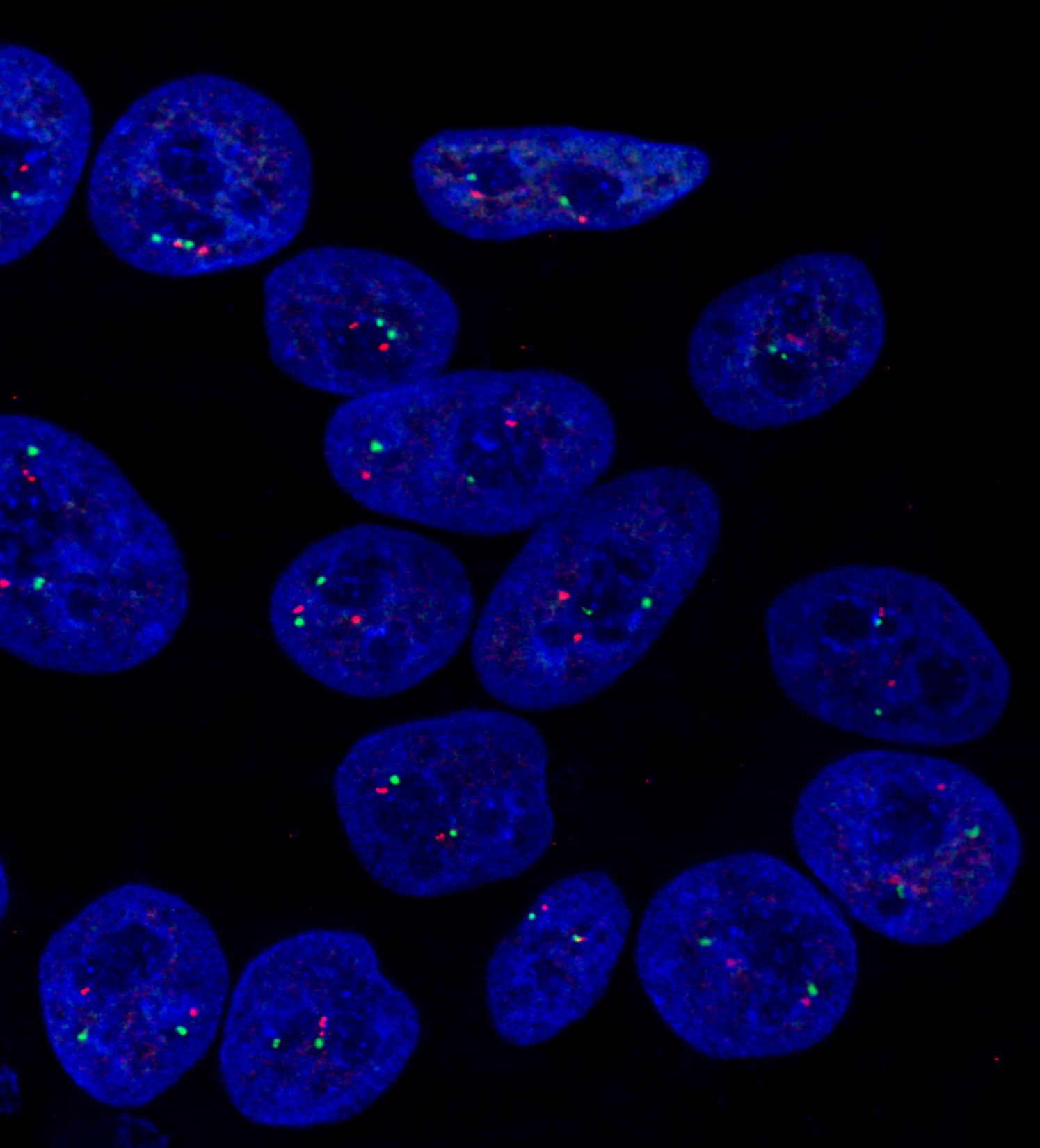
Why do hPSCs acquire karyotypic abnormalities over time and how can researchers conducting longer-term studies combat this?
AH: The mechanisms behind karyotypic instability are poorly understood. Cytogenetic changes observed in hPSCs range from whole and partial chromosomal gains, losses and translocations to sub-microscopic structural variants and single base pair changes. There are a number of potential causes of genetic variation, including errors in mitosis, sister chromatid segregation, and DNA replication, damage and repair pathways. Several researchers have given great webinars on these topics if you want to know more.
Awareness is key in conducting long-term studies. It is widely accepted that hPSCs acquire karyotypic changes. They can happen spontaneously even in carefully maintained cultures, so no one should feel embarrassed or try to hide abnormalities. Reporting any cytogenetic changes and their effects on cell behavior helps everyone in the field. That said, prolonged culture of hPSCs constantly selects for cells having improved survival or proliferative capacities. Researchers should try to avoid stressful situations such as single cell seeding or “rescuing” poor cultures that may create opportunities for variant cells.
What should researchers consider when selecting their cell culture system?
MK: Consider which particular features will benefit your experimental workflow firstly from a scientific perspective. For example, a low-protein medium like TeSR™-E8™ could be a huge advantage in a proteomic study where high levels of albumin could make analysis difficult. If accessing a selection of compatible differentiation protocols is important, mTeSR™1 has been used in >2000 publications and many differentiation kits are commercially available.
Then consider whether your application will be research use only or if particular features like a good manufacturing practice (GMP) or animal component-free medium will be required. Finally, consider if there are other workflow or scheduling needs for the lab. If feeding schedule versatility is required to work around classes or weekends, a stabilized medium such as mTeSR™ Plus could be a good option.
Does changing to a new cell culture medium affect cell quality, and can this be done easily?
MK: I think this depends on the conditions, since an adaptation period could create cell stress or a survival bottleneck. In our experience, when switching between relatively similar conditions such as different TeSR™ media, small protocol tweaks may be required but cells should not undergo adaptation or selection. An example that we perform routinely is adjusting the split ratios if you’re seeing different plating efficiencies or a faster growth rate in mTeSR™ Plus compared to in mTeSR™1. We recommend carrying cultures in parallel until those in the new medium are fully established. This allows for easy detection of changes in cell quality characteristics compared to the previous conditions.
How could the needs of researchers culturing cells change in the future?
MK: As the number of banked iPS cell lines continues to grow, researchers will want to decrease their cell maintenance labor with simplified protocols and reduced feeding requirements. We recently hosted a webinar with the Cedars-Sinai Stem Cell Core (LA, USA) describing their experience using mTeSR™ Plus medium to facilitate this. In addition, researchers will need consistent workflows that work robustly across different cell lines in order to compare the lines in differentiation or disease modelling studies. Of course, researchers moving towards pre-clinical work will need communication and supply chain support from their reagent providers to ensure that their compliance needs can be met, including any required documentation.
AH: There is a renewed focus on cell quality in the hPSC field and there are initiatives working with key stakeholders to reach a consensus on what assessments are important and what characterization should be required. STEMCELL recently partnered with Nature Research and a panel of experts to discuss current challenges and best practices for hPSC quality. Once a consensus becomes widely accepted, it is likely that journals will require similar information before publication, and the technologies available for quality assessment will evolve with this demand.