Cell therapy weekly: Leucid Bio and Lonza collaborate on new platform for manufacturing of personalized CAR-T cells
This week: Leucid Bio and Lonza collaborate on a new platform for the manufacturing of personalized CAR-T cells, Help Therapeutics completes US$25 million financing to develop iPSC heart cell injection, Sigilon Therapeutics announces update on their SIG-001 study after being put on hold by the FDA, and Quell Therapeutics raises US$156 million to advance its cell therapy pipeline.
The news highlights:
- Leucid Bio and Lonza collaborate on new platform for manufacturing of personalized CAR-T cells
- Help Therapeutics completes US$25 million to develop iPSC heart injection
- Sigilon Therapeutics announces update on SIG-001 study after being put on hold by FDA
- Quell Therapeutics raises US$156 million to advance cell therapy pipeline
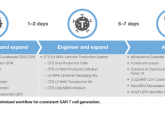
Leucid Bio and Lonza collaborate on new platform for manufacturing of personalized CAR–T cells
Biotech company Leucid Bio (London, UK) has entered a strategic collaboration with Lonza (Basel, Switzerland) to utilize Lonza’s Cocoon® Platform to optimize the automated manufacturing process for personalized CAR-T cell therapies.
Through the partnership, both companies will aim to deliver high-quality and cost-effective cell therapies in a decentralized manufacturing model. The platform will bolster Leucid Bio’s Phase I/II clinical studies with its lead candidate, LEU-011, for the treatment of platinum-resistant ovarian cancer.
John Maher, Chief Scientific Officer of Leucid Bio, said: “This is a fantastic opportunity to collaborate with Lonza, enabling us to develop a state-of-the-art scalable manufacturing process. This will significantly help Leucid to efficiently transition our CAR-T pipeline for maximum patient benefit.”
Help Therapeutics completes US$25 million financing to develop iPSC heart cell injection
iPSC therapeutics company Help Therapeutics (Nanjing, China) has announced the completion of a Series C financing round of US$25 million.
The proceeds will help the company conduct and complete clinical trials of its property development pipeline, induced pluripotent stem cell (iPSC) heart cell injection, and will initiate clinical studies of immune cell therapy based on iPSC technology.
The funding will also help the company advance its work in manufacturing cell therapy using iPSC technology focused on the fields of heart failure and tumor immune diseases.
Sigilon Therapeutics announces update on SIG-001 study after being put on hold by FDA
Biotechnology company Sigilon Therapeutics (MA, USA) has reported that fibrosed spheres were observed during a retrieval procedure in a patient in its Phase 1/2 study of SIG-001 in hemophilia A.
The FDA had placed the SIG-001 trial, investigating Sigilon’s proprietary Shielded Living Therapeutics™ cell-based therapeutics, on hold in July 2021 following their submission of an adverse event report relating to the development of inhibitors to Factor VIII in one of three patients treated.
However, the recent findings, determining that the spheres placed in the patient had fibrosed and that cells within the spheres were no longer available, have brought the trial’s significance to light.
“Patients’ safety and welfare are our highest priority. We are gathering information in order to understand these observations,” said Rogerio Vivaldi, President and CEO of Sigilon. “We will be working closely with the FDA, other regulators, and advisors to determine the impact of these observations on our programs.”
The status of the clinical hold investigation, and the findings, will be reviewed by the Safety Review Committee for SIG-001 at its next meeting in December.
Quell Therapeutics raises US$156 million to advance cell therapy pipeline
Developer of engineered T-regulatory (Treg) cell therapies Quell Therapeutics (London, UK) has announced it has raised US$156 million in an oversubscribed Series B funding.
The funding will help Quell advance its multi-modular engineered T regulatory (Treg) cell therapy pipeline and platform. It will also be used to fund the LIBERATE Phase 1/2 clinical trial of QEL-001, an antigen-specific multi-modular CAR-Treg cell therapy candidate designed to prevent rejection in liver transplant patients. In addition to this, the funding will advance the company’s pipeline in core therapeutics areas of transplantation, neuroinflammatory diseases and autoimmune diseases.
“Quell is at the forefront of a new wave of cell therapy. We are leading the way with our highly-differentiated, multi-modular approach to Treg therapy engineering and production,” said Iain McGill, Chief Executive Officer of Quell Therapeutics. “We are proud to have the support of this premiere syndicate of investors as we drive forward to our next stage of growth. With this financing, we have the full suite of capabilities – capital, cutting-edge science, and a world-class team – to advance our pipeline and platform to key milestones on our path ultimately to deliver potentially transformative therapies to patients suffering from diseases caused by immune dysregulation.”
You might also like: