Contracting organoids containing innervated muscle created

Functional neuromuscular organoids with the ability to self-organize into muscle tissue and spinal cord neurons have been developed by a research team from the Max Delbrück Center for Molecular Medicine (Berlin, Germany).
For the past decade it has been a persistent challenge to simultaneously grow two different tissue types into a single functioning organoid; however, a research team from the Max Delbrück Center for Molecular Medicine (Berlin, Germany) have developed functional neuromuscular organoids able to self-organize into muscle tissue and spinal cord neurons. Published in Cell Stem Cell, the team demonstrated the organoid’s ability to form a complex neuronal network that leads muscle tissue to contract.
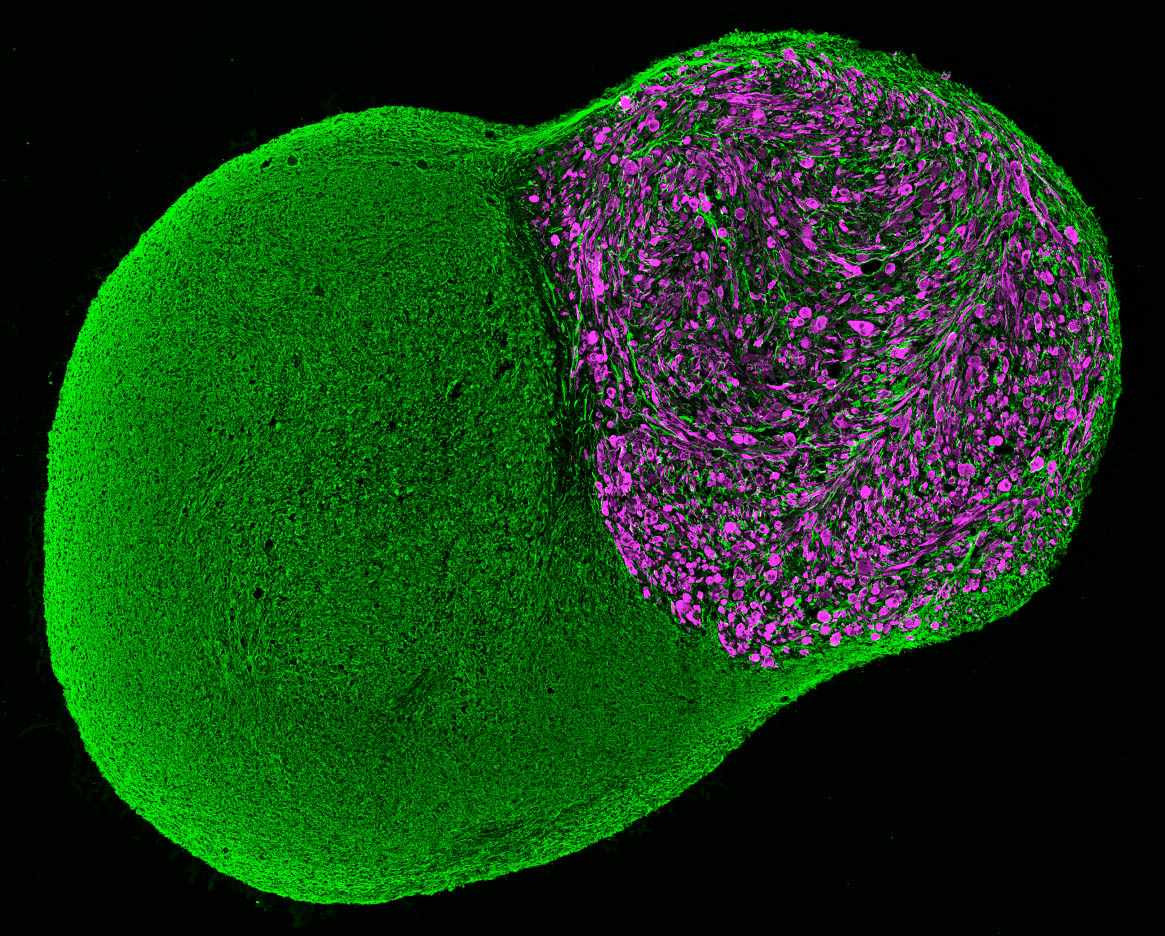
The research team placed axial stem cells, which are capable of differentiating into both spinal cord and skeletal muscle cells, into a 3D cell culture and found that the cells differentiated and self-organized into complex structures. Impressively, these cells developed functional neuromuscular junctions and complex spinal neural networks similar to central pattern generators that allow for oscillating signals and coordinated movements, such as walking and breathing.
“Our initial goal was to develop functional neuromuscular junctions, but the findings exceeded our expectations because the additional development of the CPG [central pattern generator]-like networks was an exciting but unexpected finding,” explained Mina Gouti, (Max Delbrück Center). “This has not been shown in a human in vitro model before, and offers entirely novel possibilities, including the study of CPG involvement in neurodegenerative diseases.”
The neuromuscular organoids can be kept alive for several months without deteriorating, with the oldest currently lasting 1.5 years, and can be produced from various human pluripotent stem cell lines with similar efficiency, making this a widely applicable approach to be utilized in the research of neuromuscular diseases.
“It is striking that the two different tissues can self-organize in 3D and develop advanced functional networks,” commented Gouti. “These organoids started contracting after 40 days in culture. This activity was driven by the release of neurotransmitter acetylcholine from the resident motor neurons in the organoid and was not due to spontaneous muscle activity seen in other systems. We were able to show that, because pharmacological blocking of the acetylcholine receptors was sufficient to abolish muscle contraction.”
The researchers proceeded to evaluate the potential of their organoids for the study of neuromuscular diseases. Serum from two patients with the severe autoimmune condition myasthenia gravis was applied to several organoids. After 72 hours, researchers observed fewer muscle contractions, mirroring the muscle weakness that patients experience.
“The derivation of NMOs using patient-derived iPSCs will allow us to investigate their precise origin and progression. These organoids are better suited to study the contributions of specific cell types, such as terminal Schwann cells, at different stages of neuromuscular junction development and maturation that may contribute to neuromuscular diseases,” concluded Gouti.
Future research will use patient-derived neuromuscular organoids to evaluate the effectiveness of various drugs for personalized medicine approaches.
Sources: Martins J, Fischer C, Urzi A, et al. Self-organizing 3D human trunk neuromuscular organoids. Cell Stem Cell. doi:10.1016/j.stem.2019.12.007 (2019) (Epub ahead of print); www.mdc-berlin.de/news
Have any additional questions about this story? Ask us in the comments, below.
Find out more in these top picks from the Editor:
- Advanced organoids formed from human IPSCs
- Peek behind the paper: bone healing from organoids
- Organoids: ask the experts