Leading the way to individualized cystic fibrosis treatments with primary nasal cells
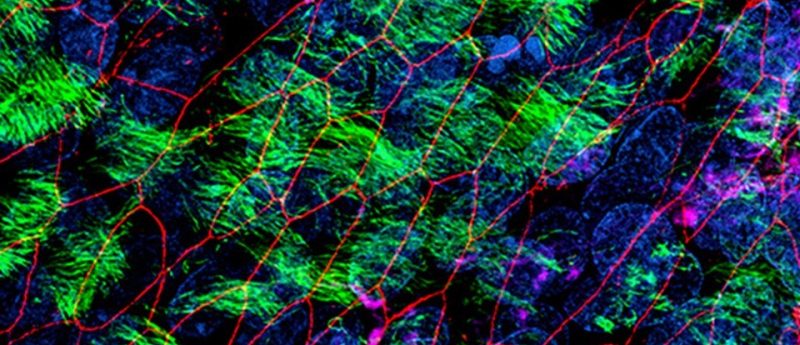
Cystic fibrosis is a genetic disease caused by a wide range of mutations in the gene encoding the CFTR protein. In this editorial, the role of primary nasal epithelial cells is explored as a valuable research model for cystic fibrosis, highlighting the broad utility of advanced culture systems.
Cystic fibrosis (CF) is a genetic disease caused by a wide range of mutations in the gene encoding the CFTR protein. Though a multisystem disease, the resulting disruption of epithelial ion and fluid transport primarily impairs the ability to clear mucus and pathogens from the airways, ultimately leading to recurrent lung infections, chronic inflammation and respiratory insufficiency [1]. The etiology of CF is relatively well understood; however, not all mutations have effective drugs available and, even for mutations with available drugs, there is high patient-to-patient variability in response to treatment. There is thus a clear need to develop tools and technologies for personalized medicine in CF [2].
Watch the full webinar and learn more about ALI cultures here>>
Enter the Cystic Fibrosis Individualized Therapy (CFIT) program, a collaboration between The Hospital for Sick Children (SickKids®) and Cystic Fibrosis Canada (both ON, Canada) with the goal of accelerating the development of precision medicine for individuals living with CF. A multidisciplinary group started in 2015, the CFIT team includes basic scientists, research nurses, clinicians and biostatisticians working collaboratively to build and validate a collection of in vitro models from CF patients. These models could then be used to develop novel treatments and personalized therapies for CF.
Theo Moraes (SickKids) recently presented a webinar on the approach that he and collaborators in CFIT are taking to study CF in vitro. In the presentation, Moraes is quick to point out that although there is no shortage of immortalized cell line models available to CF researchers, these models fall short at modeling all aspects of the disease physiology and truly reflecting the disease mechanism for individual patients. To address this gap, Moraes and the CFIT team use primary nasal epithelial cells, cultured at the air—liquid interface (ALI), to model CF and patient-specific responses to different therapeutic approaches.
The key advantages of using nasal cells are that they are easily obtained and can generate cultures representing the bronchial epithelium, including the typical CF phenotype in that lung region [2]. In his webinar, Moraes demonstrates how ALI cultures of primary nasal epithelial cells could be useful in designing optimal therapeutic approaches for individuals with CF. Nasal cells collected from a patient can be cultured and used to rank different drugs and drug combinations based on their corrective effect on epithelial function. In this way, CFIT’s bank of characterized nasal cultures could be used to accelerate the development of individualized therapies for CF patients.
Reiterating that the median survival for a CF patient in Canada is just over 50 years of age, Moraes emphasizes the need both for new drugs and drug combinations to treat CF, as well as individual patients. He underscores the utility of in vitro cultures derived from primary nasal epithelial cells for screening new drugs on multiple patients during drug development, as well as testing multiple therapeutic approaches for the same patient. Moving beyond small molecule therapeutics, Moraes describes how these in vitro systems can also be used to model gene delivery or editing strategies. These applications for ALI cultures of primary airway cells highlight the broad utility of advanced culture systems, including organoids and induced pluripotent stem cell cultures, for providing in vitro data that are predictive of in vivo physiology.
Watch the full webinar and learn more about ALI cultures here>>
References
- Ratjen F, Bell SC, Rowe SM, Goss CH, Quittner AL, Bush A. Cystic fibrosis. Nat Rev Dis Primers. 1, 15010 (2015).
- Awatade NT, Wong SL, Hewson CK et al. Human primary epithelial cell models: promising tools in the era of cystic fibrosis personalized medicine. Front Pharmacol. 9,1429 (2018).