Scorpion venom repurposed for CAR-T therapy

CAR-T cells engineered with a component of scorpion venom have been found to improve the selectivity of this therapy for glioblastoma (GBM).
Scientists from City of Hope (CA, USA) have incorporated a component of scorpion venom into the binding domain of engineered CAR-T cells. The isolate peptide sequence, chlorotoxin (CLTX), enables CAR-T cells to recognise and kill tumor cells — providing a new avenue of therapy for certain types of brain cancer.
“Much like a scorpion uses toxin components of its venom to target and kill its prey, we’re using chlorotoxin to direct the T cells to target the tumor cells with the added advantage that the CLTX-CAR-T cells are mobile and actively surveilling the brain looking for appropriate targets,” commented Michael Barish, City of Hope Professor and Chair of the Department of Developmental and Stem Cell Biology.
In their study, recently published in Science Translational Medicine, the novel CAR-T cells were engineered with the scorpion venom to target glioblastoma (GBM) – one of the deadliest types of cancer – and one of the most common forms of brain cancer.
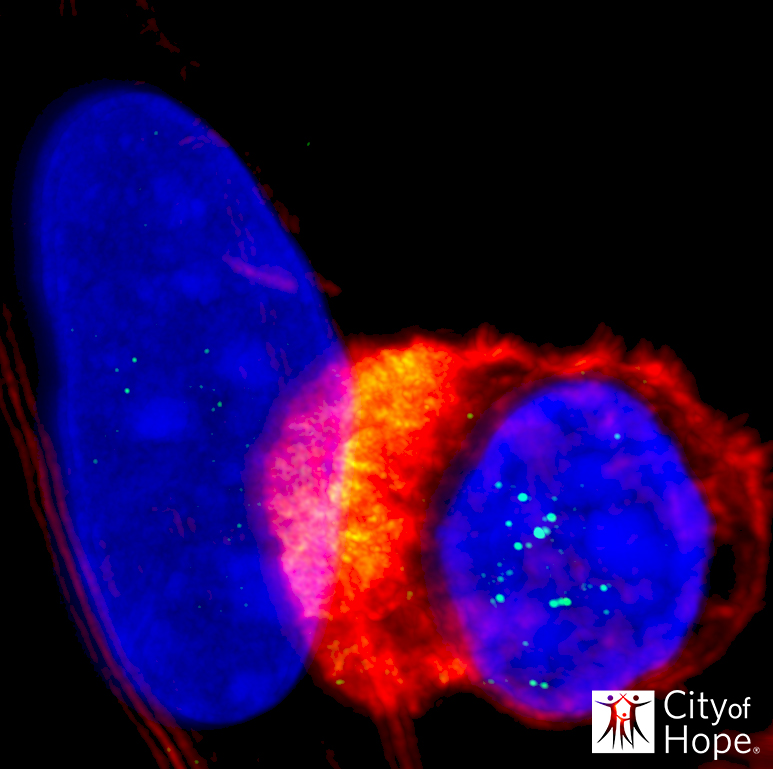
The team found that the scorpion sequence, CLTX, can successfully bind to a high proportion of GBM cells within tumor samples from patients — implying that this toxin can be used to improve tumor specific binding and activation of CAR-T cells.
“We are not actually injecting a toxin, but exploiting CLTX’s binding properties in the design of the CAR. The idea was to develop a CAR that would target T cells to a wider variety of GBM tumor cells than the other antibody-based CARs,” Barish explained.
The CLTX-CAR-T cells were able to wipe out a large population of GBM cell lines, whilst leaving non-tumor cells in the brain and other organs unharmed. What’s more, the team think that CLTX can also target GBM stem-like cells which are thought to enable tumor recurrence.
“Our chlorotoxin-incorporating CAR expands the populations of solid tumors potentially targeted by CAR-T cell therapy, which is particularly needed for patients with cancers that are difficult to treat such as glioblastoma,” commented Christine Brown, City of Hope’s Heritage Provider Network Professor in Immunotherapy and deputy director of T Cell Therapeutics Research Laboratory.
“This is a completely new targeting strategy for CAR-T therapy with CARs incorporating a recognition structure different from other CARs,” Brown continued.
The team hopes to develop this therapy further, with the first-in-human clinical trials now screening for patients.
Sources: Wang D, Starr R, Chnag WC et al. Chlorotoxin-directed CAR T cells for specific and effective targeting of glioblastoma. Sci. Transl. Med. 12(533), eaaw2672 (2020); www.eurekalert.org/pub_releases/2020-03/coh-fst022720.php
Find out more in these top picks from the Editor:
- Could light sensitive CAR-T cells treat skin cancers?
- How NICE view advanced therapies: an interview with Pilar Pinilla DomÃnguez
- Cell therapy weekly: UltraCAR-T given boost in fight against acute myeloid leukemia