Understanding umbilical cord blood: an interview with Chris Jones
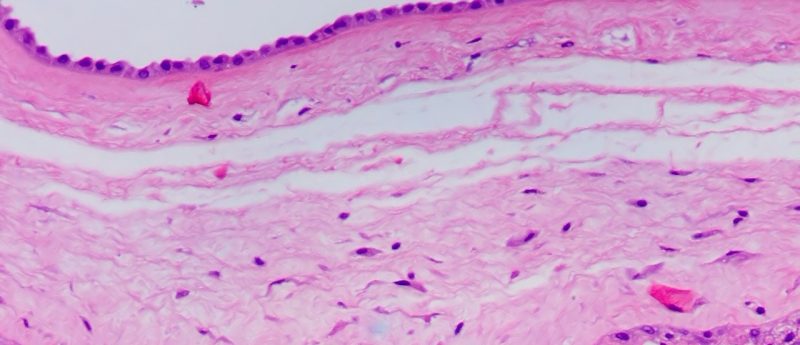
In this interview, Chris Jones (Burst Biologics) discusses the promising field of orthobiologics, particularly umbilical cord blood for bone repair. Please introduce yourself and Burst Biologics. My name is Chris Jones. I’m the founder and CEO of Burst Biologics (ID, USA), which was founded in 2010 under the name of Smart Surgical. We set out with the focus to develop innovative technologies in the orthobiologics space. What prompted Burst Biologics to do this research on bone repair and cytokines? As an organization, we had a very strong area of interest in orthobiologics, particularly bone fusions in spinal segments. We understood...