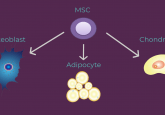
Spotlight on MSCs
MSCs can be harvested from several human tissues, or derived from induced pluripotent stem cells (iPSCs), and can differentiate into a range of tissue-specific cells. Various studies have also shown the MSCs, unlike iPSCs, show few oncogenic properties, potentially making them safer as therapeutic agents. Their strong capacity for differentiation also makes MSCs useful as a tool to model disease progression and assess drug efficacy. The spotlight will aim to review the current challenges in the production of MSCs with a focus on consistency, safety and efficacy. We will also look into the clinical applications of MSCs for regenerative medicine and how these therapies are going to evolve and change healthcare in the coming years.